- Introduction to virology -1
- Introduction to virology – 2
- Virology tables
- DNA enveloped viruses
- DNA non-enveloped viruses
- RNA enveloped viruses (orthomyxoviruses, paramyxoviruses, coronaviruses, togaviruses, rhabdoviruses, retroviruses)
- RNA non-enveloped viruses
- Hepatitis viruses
- Arboviruses (including HIV)
- Viruses classified according to target organ
All posts by DentMistry
Gross Anatomy
Viruses classified according to target organ
Below are the following topics that are listed in table format:
- Viruses classified according to their nuclear structure
- Diseases caused by viruses commonly infecting the intestinal tracts
- Viruses that cause hemorrhagic fevers and their vectors
- Human tumor viruses
- Features of viruses that infect the respiratory tract: DNA and RNA Viruses
- Positive sense viral RNA
- Negative sense Viral RNA


Arboviruses (Including HIV)
All arboviruses are transmitted by arthropods (arthropodborne) such as mosquitoes and ticks from the wild animal reservoir to humans.
FLAVIVIRUS
Transmission – For effective transmission to occur, the virus must be present in the bloodstream of the vertebrate host (viremia) in sufficiently high titer to be taken up in the small volume of blood ingested during an insect bite. After ingestion, the virus replicates in the gut of the arthropod and then spreads to other organs, including the salivary glands. Only the female of the species serves as the vector of the virus, because only she requires a blood meal in order for progeny to be produced. An obligatory length of time, called the extrinsic incubation period, must pass before the virus has replicated sufficiently for the saliva of the vector to contain enough virus to transmit an infectious dose.
For most viruses, the extrinsic incubation period ranges from 7 to 14 days.
Clinical Findings – (1) encephalitis; (2) hemorrhagic fever or (3) fever with myalgias, arthralgias, and nonhemorrhagic rash. Immunity is usually lifelong

Yellow Fever Virus
Member of the flavivirus family. Causes yellow fever in the tropical areas of Africa and South America. “Jungle” yellow fever is transmitted from monkeys to humans by mosquitoes.
“Urban” yellow fever is transmitted from human to human by Aedes mosquitoes (i.e., humans are the reservoir in the urban form). Humans are not a “dead-end” host because viremia is high.
Dengue Virus
Member of the flavivirus family.
Transmission – by Aedes mosquitoes from one human to another. A monkey reservoir is suspected.
Second episodes may result in dengue hemorrhagic fever, a life-threatening complication.
The pathogenesis is as follows: The patient recovers from classic dengue caused by one of the four serotypes, and antibody against that serotype is produced. When the patient is infected with another serotype of dengue virus, an anamnestic, heterotypic response occurs, and large amounts of cross-reacting antibody to the first serotype are produced. There are two hypotheses about what happens next. One is that immune complexes composed of virus and antibody are formed that activate complement, causing increased vascular permeability and thrombocytopenia. The other is that the antibodies increase the entry of virus into monocytes and macrophages, with the consequent liberation of a large amount of cytokines. In either scenario, shock and hemorrhage result.
HUMAN IMMUNODEFICIENCY VIRUS
Characteristics—Enveloped virus with two copies (diploid) of a single-stranded, positive-polarity RNA genome. RNAdependent DNA polymerase (reverse transcriptase) makes a DNA copy of the genome, which integrates into host cell DNA. Precursor polypeptides must be cleaved by virus encoded protease to produce functional viral proteins. The tat gene encodes a protein that activates viral transcription. Antigenicity of the gp120 protein changes rapidly; therefore, there are many serotypes.
HIV preferentially infects and kills helper (CD4) T lymphocytes, resulting in the loss of cell-mediated immunity and a high probability that the host will develop opportunistic infections.

There are several important antigens of HIV:
(1) gp120 and gp41 are the type-specific envelope glycoproteins. gp120 protrudes from the surface and interacts with the CD4 receptor (and a second protein, a chemokine receptor) on the cell surface. gp41 is embedded in the envelope and mediates the fusion of the viral envelope with the cell membrane at the time of infection. The gene that encodes gp120 mutates rapidly, resulting in many antigenic variants.
(2) The group-specific antigen, p24, is located in the core and is not known to vary. Antibodies against p24 do not neutralize HIV infectivity but serve as important serologic markers of infection.
Summary of Replicative Cycle
- Entry of HIV into the cell is the binding of the virion gp120 envelope protein to the CD4 protein on the cell surface. The virion gp120 protein then interacts with a second protein on the cell surface, one of the chemokine receptors.
- Next, the virion gp41 protein mediates fusion of the viral envelope with the cell membrane, and the virion core containing the nucleocapsid, RNA genome, reverse transcriptase, protease and intergrase enzyme enters the cytoplasm.
- Chemokine receptors, such as CXCR4 and CCR5 proteins, are required for the entry of HIV into CD4-positive cells.
- Mutations in the gene encoding CCR5 endow the individual with protection from infection with HIV. People who are homozygotes are completely resistant to infection, and heterozygotes progress to disease more slowly.
- In the cytoplasm, reverse transcriptase transcribes the genome RNA into double-stranded DNA, which migrates to the nucleus, where it integrates into the host cell DNA.
- The viral DNA can integrate at different sites in the host cell DNA, and multiple copies of viral DNA can integrate.
- Integration is mediated by a virus-encoded endonuclease (integrase).
- Viral mRNA is transcribed from the proviral DNA by host cell RNA polymerase (augmented by virus encoded Tat protein) and translated into several large polyproteins.
- The Gag and Pol polyproteins are cleaved by the viral protease, whereas the Env polyprotein is cleaved by a cellular protease.
- The Gag polyprotein is cleaved to form the main core protein (p24), the matrix protein (p17), and several smaller proteins. The Pol polyprotein is cleaved to form the reverse transcriptase, integrase, and protease.
- Cleavage by the viral protease occurs as the immature virion buds from the cell membrane.
- It is this cleavage process that results in the mature, infectious virion.
Note that HIV replication is dependent on cell proteins as well as viral proteins. First there are the cell proteins required during the early events, namely CD4, and the chemokine receptors, CCR5 and CXCR4. Cell proteins, such as actin and tubulin, are involved with the movement of viral DNA into the nucleus. The cell protein cyclin T1 and the viral protein Tat are part of the complex that transcribes viral mRNA. Cell proteins are also involved in the budding process by which the virus exits the cell.
Pathogenesis—
HIV is first found in the blood 4 to 11 days after infection.
Two receptors are required for HIV to enter cells. One receptor is CD4 protein found primarily on helper T cells. HIV infects and kills helper T cells, which predisposes to opportunistic infections. Other cells bearing CD4 proteins on the surface (e.g., astrocytes) are infected also. The other receptor for HIV is a chemokine receptor such as CCR5. The NEF protein is an important virulence factor. It reduces class I MHC protein synthesis, thereby reducing the ability of cytotoxic T cells to kill HIV-infected cells.
The main immune response to HIV infection consists of cytotoxic CD8-positive lymphocytes. These cells respond to the initial infection and control it for many years. It is the ultimate failure of these cytotoxic T cells that results in the clinical picture of AIDS. Cytotoxic T cells lose their effectiveness because so many CD4 helper T cells have died; thus the supply of lymphokines, such as IL-2, required to activate the cytotoxic T cells is no longer sufficient.
HIV has three main mechanisms by which it evades the immune system: (1) integration of viral DNA into host cell DNA, resulting in a persistent infection; (2) a high rate of mutation of the env gene; and (3) the production of the Tat and Nef proteins that downregulate class I MHC proteins required for cytotoxic T cells to recognize and kill HIVinfected cells. The ability of HIV to infect and kill CD4- positive helper T cells further enhances its capacity to avoid destruction by the immune system.

Hepatitis Virus – Hep A, B, C, D, E
Main site of infection is the liver. These five are hepatitis A virus (HAV), hepatitis B virus (HBV), hepatitis C virus (HCV), hepatitis D virus (HDV, delta virus), and hepatitis E virus (HEV)

Hepatitis A Virus
Summary of Replicative Cycle – HAV has a replicative cycle similar to that of poliovirus
Pathogenesis—The virus replicates in the GI tract and then spreads to the liver during a brief viremic period. The virus is not cytopathic (does not harm) for the hepatocyte. Hepatocellular injury is caused by immune attack by cytotoxic T cells. Virus excreted in the stools for 2 weeks preceding onset of symptoms
Hepatitis B Virus
Characteristics—Incomplete circular double-stranded DNA (i.e., one strand has about one-third missing and the other strand is “nicked” [not covalently bonded]). DNA polymerase in virion. HBV-encoded polymerase acts as a reverse transcriptase by using viral mRNA as the template for the synthesis of progeny genome DNA.
There are three important antigens: the surface antigen(HBsAg), the core antigen (HBcAg) – The core antigen forms the nucleocapsid core of the virion, and the e antigen (HBeAg)- is secreted from infected cells into the blood. The e antigen is an important indicator of transmissibility. In the patient’s serum, long rods and spherical forms composed solely of HBsAg predominate. HBV has one serotype based on the surface antigen.
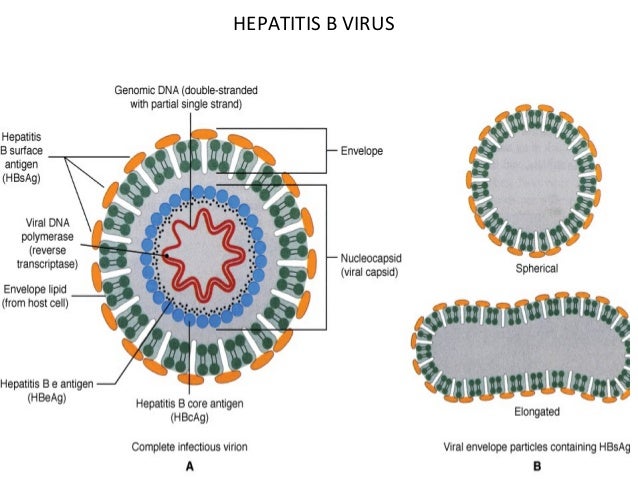
Summary of Replicative Cycle
- After entry of the virion into the cell and its uncoating
- Carries Reverse Transcriptase enzyme that completes the single stranded DNA portion to form double strand
- The virion DNA polymerase synthesizes the missing portion of DNA, and a double-stranded closed-circular DNA is formed in the nucleus.
- This DNA serves as a template for mRNA synthesis by cellular RNA polymerase.
- After the individual mRNAs are made, a full-length positive-strand transcript is made, which is the template for the minus strand of the progeny DNA.
- The minus strand then serves as the template for the plus strand of the genome DNA.
- This RNA-dependent DNA synthesis catalyzed by reverse transcriptase encoded by HBV takes place within the newly assembled virion core in the cytoplasm.
- The RNAdependent DNA synthesis that produces the genome and the DNA-dependent DNA synthesis that fills in the missing portion of DNA soon after infection of the next cell are carried out by the same enzyme (i.e., the HBV genome encodes only one polymerase).
- Hepadnaviruses are the only viruses that produce genome DNA by reverse transcription with mRNA as the template. (Note that this type of RNA-dependent DNA synthesis is similar to but different from the process in retroviruses, in which the genome RNA is transcribed into a DNA intermediate.)
- Some of the progeny DNA integrates into the host cell genome, and this seems likely to be the DNA that maintains the carrier state.
- Progeny HBV with its HBsAg-containing envelope is released from the cell by budding through the cell membrane.
NB: There is a high incidence of hepatocellular carcinoma (hepatoma) in many Asian countries—a finding that indicates that HBV may be a human tumor virus. It appears that the HBV vaccine is the first vaccine to prevent a human cancer.
Pathogenesis—Hepatocellular injury due to immune attack by cytotoxic (CD8) T cells. Antigen–antibody complexes cause arthritis, rash, and glomerulonephritis. About 5% of HBV infections result in a chronic carrier state. Chronic hepatitis, cirrhosis, and hepatocellular carcinoma can occur. Hepatocellular carcinoma may be related to the integration of part of the viral DNA into hepatocyte DNA.
Virus enters blood circulation – infects epatocytes – viral antigens displayed on hepatocytes – cytotoxic T cells mediate an immune attack against the viral antigens – inflammation and necrosis occurs
Clinical Findings
- Many HBV infections are asymptomatic and are detected only by the presence of antibody to HBsAg.
- The incubation period for hepatitis B is 10 to 12 weeks, which is much longer than that of hepatitis A (3–4 weeks).
- Compared to HAV, hepatitis B, symptoms tend to be more severe, and life-threatening hepatitis can occur.
- Patients coinfected with both HBV and human immunodeficiency virus (HIV) may have increased hepatic damage if HIV is treated prior to treating HBV. This occurs because the “immune reconstitution” that results when HIV is treated successfully leads to increased damage to the hepatocytes by the restored, competent cytotoxic T cells. For this reason, it is suggested that HBV be treated prior to treating HIV.
Hepatitis C Virus
Summary of Replicative Cycle – The replication of HCV is like other flaviviruses
Pathogenesis—Hepatocellular injury caused by cytotoxic T cells. HCV replication itself does not kill cells (i.e., does not cause a cytopathic effect). More than 50% of infections result in the chronic carrier state. The chronic carrier state predisposes to chronic hepatitis and to hepatocellular carcinoma.
Hepatitis D Virus
Characteristics—Defective virus that uses hepatitis B surface antigen as its protein coat. HDV can replicate only in cells already infected with HBV (i.e., HBV is a helper virus for HDV). HDV has one serotype (because HBV has only one serotype).
Pathogenesis—Hepatocellular injury probably caused by cytotoxic T cells. Chronic hepatitis and chronic carrier state occur.
Hepatitis E Virus
Causes outbreaks of hepatitis, primarily in developing countries. Similar to hepatitis A virus in the following ways: transmitted by fecal–oral route, no chronic carrier state, no cirrhosis, and no hepatocellular carcinoma. No antiviral therapy and no vaccine. HEV is a nonenveloped, single-stranded RNA virus
RNA Non-Enveloped Viruses
PICORNAVIRUSES
The genome RNA is unusual because it has a protein on the 5′ end that serves as a primer for transcription by RNA polymerase. Picornaviruses replicate in the cytoplasm of cells. The picornavirus includes two groups of medical importance: the enteroviruses and the rhinoviruses.
ENTEROVIRUSES
Enteroviruses infect primarily the enteric tract. Enteroviruses replicate optimally at 37°C. Enteroviruses are stable under acid conditions (pH 3–5), which enables them to survive exposure to gastric acid
Poliovirus
Characteristics—Genome RNA acts as mRNA and is translated into one large polypeptide, which is cleaved by virus– encoded protease to form functional viral proteins.
Summary of Replicative Cycle
- The virion interacts with specific cell receptors on the cell membrane and then enters the cell.
- After uncoating, the genome RNA functions as mRNA and is translated into one very large polypeptide called noncapsid viral protein 00.
- This polypeptide is cleaved by a virus-encoded protease in multiple steps to form both the capsid proteins of the progeny virions and several noncapsid proteins, including the RNA polymerase that synthesizes the progeny RNA genomes.
- Replication of the genome occurs by synthesis of a complementary negative strand, which then serves as the template for the positive strands.
- Some of these positive strands function as mRNA to make more viral proteins, and the remainder become progeny virion genome RNA.
- Assembly of the progeny virions occurs by coating of the genome RNA with capsid proteins.
- Virions accumulate in the cell cytoplasm and are released upon death of the cell. They do not bud from the cell membrane.
Pathogenesis—The virus replicates in the pharynx and the GI tract. It can spread to the local lymph nodes and then through the bloodstream to the central nervous system. Most infections are asymptomatic or very mild. Aseptic meningitis is more frequent than paralytic polio. Paralysis is the result of death of motor neurons, especially anterior horn cells in the spinal cord. Pathogenesis of postpolio syndrome is unknown.
Coxsackie Viruses
Summary of Replicative Cycle – Replication is similar to that of poliovirus.
Pathogenesis—The initial site of infection is the oropharynx, but the main site is the GI tract. The virus spreads through the bloodstream to various organs.
| Hand, foot and mouth disease | Herpangina |
| Coxsackie A16 | Coxsackie A1-6, 8, 10, 22 |
| Airborne, feco-oral | Feco-oral, contaminated saliva |
| Children | Children |
| Fever, malaise, lymphadenopathy, sore mouth | Fever, malaise, dysphagia, sore throat |
| Oral vesicles – yellow membrane and erythematous halo, also on hand and foot | Vesicles ulcerate in soft palate and pharynx |
| Resolution in 1-2 weeks, symptomatic treatment | Self limiting |
RHINOVIRUSES
Characteristics— Rhinoviruses are found in the nose and throat. Rhinoviruses grow at 33°c, in accordance with the lower temperature of the nose. Rhinoviruses are destroyed by stomach acid and therefore do not replicate in the GI tract.
Summary of Replicative Cycle- Replication is similar to that of poliovirus. The cell surface receptor for rhinoviruses is intracellular adhesion molecule 1 (ICAM-1)
Pathogenesis—Infection is limited to the mucosa of the upper respiratory tract and conjunctiva. The virus replicates best at the low temperatures of the nose and less well at 37°C, which explains its failure to infect the lower respiratory tract.
CALICIVIRUSES
Norwalk Virus (Norovirus)
Pathogenesis—Infection is typically limited to the mucosal cells of the intestinal tract. Watery diarrhea without red cells or white cells occurs. Many infections are asymptomatic. Immunity is brief and reinfection occurs.
REOVIRUSES
Rotavirus
Characteristics— Rotavirus is resistant to stomach acid and hence can reach the small intestine. There are at least six serotypes.
Summary of Replicative Cycle
- Rotavirus attaches to the cell surface at the site of the β-adrenergic receptor.
- After entry of the virion into the cell, the RNA-dependent RNA polymerase synthesizes mRNA from each of the 11 segments within the cytoplasm.
- The 11 mRNAs are translated into the corresponding number of structural and nonstructural proteins.
- One of these, an RNA polymerase, synthesizes minus strands that will become part of the genome of the progeny virus.
- Capsid proteins form an incomplete capsid around the minus strands, and then the plus strands of the progeny genome segments are synthesized.
- The virus is released from the cytoplasm by lysis of the cell, not by budding.
Pathogenesis— Rotavirus replicates in the mucosal cells of the small intestine, resulting in the excess secretion of fluids and electrolytes into the bowel lumen. The consequent loss of salt, glucose, and water leads to diarrhea. Diarrhea is nonbloody.
RNA Enveloped Viruses
- Orthomyxoviruses
- Paramyxoviruses
- Coronaviruses
- Togaviruses
- Rhabdoviruses
- Retroviruses
ORTHOMYXOVIRUSES
Influenza Virus
Disease—
Influenza A virus causes worldwide epidemics (pandemics) of influenza, influenza B virus causes major outbreaks of influenza, and influenza C virus causes mild respiratory tract infections but does not cause outbreaks of influenza. Pandemics occur when a variant of influenza A virus that contains a new hemagglutinin against which people do not have preexisting antibodies
Characteristics—The two major antigens are the hemagglutinin (HA) and the neuraminidase (NA) on separate surface spikes. The function of the hemagglutinin is to bind to the cell surface receptor (neuraminic acid, sialic acid) to initiate infection of the cell. The neuraminidase cleaves neuraminic acid (sialic acid) to release progeny virus from the infected cell as well as degrades the protective layer of mucus in the respiratory tract
There are two types of antigenic changes:
1) Antigenic shift in these proteins as a result of reassortment of RNA segments accounts for the epidemics of influenza caused by influenza A virus. Influenza A viruses of animals are the source of the new RNA segments.
2) Antigenic drift due to mutations also contributes. The virus has many serotypes because of these antigenic shifts and drifts. The antigenicity of the internal nucleocapsid protein determines whether the virus is an A, B, or C influenza virus.
NB: Because influenza B virus is only a human virus, there is no animal source of new RNA segments. Influenza B virus therefore does not undergo antigenic shifts. It does, however, undergo enough antigenic drift . Influenza B virus has no antigens in common with influenza A virus.

Influenza viruses have both group-specific and typespecific antigens.
(1) The internal ribonucleoprotein is the group-specific antigen that distinguishes influenza A, B, and C viruses.
(2) The hemagglutinin and the neuraminidase are the type-specific antigens located on the surface. Antibody against the hemagglutinin neutralizes the infectivity of the virus (and prevents disease), whereas antibody against the group-specific antigen (which is located internally) does not. Antibody against the neuraminidase does not neutralize infectivity but does reduce disease by decreasing the amount of virus released from the infected cell
Summary of Replicative Cycle
The virus adsorbs to the cell when the viral hemagglutinin interacts with sialic acid receptors on the cell surface.
The virus then enters the cell in vesicles and uncoats within an endosome. Uncoating is facilitated by the low pH within the endosome.
Protons pass through the ion channel formed by the M2 protein into the interior of the virion. This disrupts the virion envelope and frees the nucleocapsid to enter the cytoplasm and then migrate to the nucleus where the genome RNA is transcribed.
The virion RNA polymerase transcribes the eight genome segments into eight mRNAs in the nucleus. Synthesis of the eight mRNAs occurs in the nucleus because a methylated guanosine “cap” is required. The cap is obtained from cellular nuclear RNAs in a process called “cap snatching.”
Most of the mRNAs move to the cytoplasm, where they are translated into viral proteins. Some of the viral mRNAs remain in the nucleus, where they serve as the template for the synthesis of the negative-strand RNA genomes for the progeny virions.
Two newly synthesized proteins, NP protein and matrix protein, bind to the progeny RNA genome in the nucleus, and that complex is transported to the cytoplasm.
The helical ribonucleoprotein assembles in the cytoplasm, matrix protein mediates the interaction of the nucleocapsid with the envelope, and the virion is released from the cell by budding from the outer cell membrane at the site where the hemagglutinin and neuraminidase are located.
The neuraminidase releases the virus by cleaving neuraminic acid on the cell surface at the site of the budding progeny virions.
Pathogenesis— After the virus has been inhaled, the neuraminidase degrades the protective mucus layer, allowing the virus to gain access to the cells of the upper and lower respiratory tract epithelium. The infection is limited primarily to this area because the proteases that cleave the hemagglutinin are located in the respiratory tract.
PARAMYXOVIRUSES
Paramyxoviruses contains an RNA-dependent RNA polymerase, which transcribes the negative-polarity genome into mRNA. The genome is therefore not infectious. The envelope is covered with spikes, which contain hemagglutinin, neuraminidase, or a fusion protein that causes cell fusion and, in some cases, hemolysis
Measles Virus
Summary of Replicative Cycle
- After adsorption to the cell surface via its hemagglutinin, the virus penetrates and uncoats
- Virion RNA polymerase transcribes the negative-strand genome into mrna.
- Multiple mrnas are synthesized, each of which is translated into the specific viral proteins
- The helical nucleocapsid is assembled
- The matrix protein mediates the interaction with the envelope, and the virus is released by budding from the cell membrane.
Characteristics – Vitamin A supplementation greatly reduces the severity of measles. Patients with deficient cell-mediated immunity (e.g., AIDS patients) have a severe, life-threatening disease when they contract measles.
Pathogenesis—Initial site of infection is the upper respiratory tract. Virus spreads to local lymph nodes and then via the blood to other organs, including the skin. Giant cell pneumonia and encephalitis can occur. The maculopapular rash is due to cellmediated immune attack by cytotoxic T cells on virus-infected vascular endothelial cells in the skin.
Immunity — Lifelong immunity occurs in individuals who have had the disease. Maternal antibody passes the placenta, and infants are protected during the first 6 months of life.
Mumps Virus
Summary of Replicative Cycle – Replication is similar to that of measles virus
Pathogenesis— The virus infects the upper respiratory tract and then spreads through the blood to infect the parotid glands, testes, ovaries, pancreas, and, in some cases, meninges. Alternatively, the virus may ascend from the buccal mucosa up Stensen’s duct to the parotid gland. Lifelong immunity occurs in persons who have had the disease. Maternal antibody passes the placenta and provides protection during the first 6 months of life.
Respiratory Syncytial Virus
Characteristics—Unlike other paramyxoviruses, it has only a fusion protein in its surface spikes. The fusion protein causes cells to fuse, forming multinucleated giant cells (syncytia). It has no hemagglutinin.
Summary of Replicative Cycle – Replication is similar to that of measles virus
Pathogenesis—Infection involves primarily the lower respiratory tract in infants and is localized to the respiratory tract. Multinucleated giant cells caused by the viral fusion protein are a hallmark. Maternal antibody passed to the infant may react with the virus, form immune complexes, and damage the respiratory tract cells
Parainfluenza Virus
Characteristics—surface spikes consist of hemagglutinin (H), neuraminidase (N), and fusion (F) proteins. Unlike influenza viruses, the antigenicity of its hemagglutinin and neuraminidase is stable.
Summary of Replicative Cycle – Replication is similar to that of measles virus
Pathogenesis— These viruses cause upper and lower respiratory tract disease without viremia (virus in blood). Parainfluenza viruses 1 and 2 are major causes of croup.
CORONAVIRUSES
Characteristics—The receptor for the SARS coronavirus on the surface of cells is angiotensin converting enzyme-2.
Summary of Replicative Cycle
- The virus adsorbs to cells via its surface spikes (hemagglutinin), after which it enters the cytoplasm, where it is uncoated.
- The positive-strand genome is translated into two large polypeptides, which are self-cleaved by the virus-encoded protease.
- Two of these peptides aggregate to form the RNA polymerase that replicates the genome.
- In addition, mRNAs are synthesized, and then translated into the structural proteins.
- The virus is assembled and obtains its envelope from the endoplasmic reticulum. Replication occurs in the cytoplasm.
Pathogenesis—Infection is typically limited to the mucosal cells of the respiratory tract. At least 50% of infections are asymptomatic. Immunity is brief and reinfection occurs. Pneumonia caused by SARS coronavirus is characterized by diffuse edema in alveolar space resulting in hypoxia.
Update on: COVID – 19

TOGAVIRUSES
Rubella Virus
Characteristics—Its surface spikes contain hemagglutinin
Summary of Replicative Cycle
- After penetration of the cell and uncoating, the plus-strand RNA genome is translated into several nonstructural and structural proteins.
- One of the nonstructural rubella proteins is an RNA-dependent RNA polymerase, which replicates the genome first by making a minus-strand template and then, from that, plus strand progeny.
- Both replication and assembly occur in the cytoplasm, and the envelope is acquired from the outer membrane as the virion exits the cell.
Pathogenesis—The initial site of infection is the nasopharynx, from which it spreads to local lymph nodes. It then disseminates to the skin via the bloodstream. The rash is attributed to both viral replication and immune injury. During maternal infection, the virus replicates in the placenta and then spreads to fetal tissue. If infection occurs during the first trimester, a high frequency of congenital malformations occurs. Maternal antibody protects against fetal infection. Natural infection leads to lifelong immunity.
RHABDOVIRUSES
Rabies Virus
Summary of Replicative Cycle
- Rabies virus attaches to the acetylcholine receptor on the cell surface.
- After entry into the cell, the virion RNA polymerase synthesizes five mRNAs that code for viral proteins.
- After replication of the genome viral RNA by a virus encoded RNA polymerase, progeny RNA is assembled
- with virion proteins to form the nucleocapsid, and the envelope is acquired as the virion buds through the cell membrane.
Pathogenesis—Viral receptor is the acetylcholine receptor. Replication of virus at the site of the bite, followed by axonal transport up the nerve to the central nervous system. After replicating in the brain, the virus migrates peripherally to the salivary glands, where it enters the saliva. When the animal is in the agitated state as a result of encephalitis, virus in the saliva can be transmitted via a bite.
Within the central nervous system, encephalitis develops, with the death of neurons and demyelination. Infected neurons contain an eosinophilic cytoplasmic inclusion called a Negri body, which is important in laboratory diagnosis of rabies
Negri body:

Prevention—Preexposure prevention of rabies consists of the vaccine only.
Postexposure prevention consists of:
(1) Washing the wound
(2) Giving rabies immune globulins (passive immunization), mostly into the wound
(3) Giving the inactivated vaccine (active immunization) made in human cell culture. The decision to give the immune serum and the vaccine depends on the circumstances.
As much as possible of the RIG is given into the bite site, and the remainder is given intramuscularly.
Prevention of rabies in dogs and cats by using a killed vaccine has reduced human rabies significantly.
RETROVIRUSES
HUMAN T CELL LYMPHOTROPIC VIRUS
There are two important human retroviruses: human T-cell lymphotropic virus, which is described here, and human immunodeficiency virus (HIV)
Important Properties
- The virions of both HTLV and HIV contain a reverse transcriptase, integrase, and protease.
- The envelope proteins of HTLV are gp46 and gp21, whereas those of HIV are gp120 and gp41.
- However, HTLV does not kill T cells, whereas HIV does. HTLV causes malignant transformation that “immortalizes” the infected T cells and allows them to proliferate in an uncontrolled manner.
Summary of Replicative Cycle
- HTLV primarily infects CD4-positive T lymphocytes.
- Within the cytoplasm, reverse transcriptase synthesizes a DNA copy of the genome, which migrates to the nucleus and integrates into cell DNA.
- Viral mRNA is made by host cell RNA polymerase and transcription is upregulated by Tax protein.
- The Rex protein controls the synthesis of the gag/pol mRNA, the env mRNA, and their subsequent transport to the cytoplasm, where they are translated into structural viral proteins.
- Full-length RNA destined to become progeny genome RNA is also synthesized and transported to the cytoplasm.
- The virion nucleocapsid is assembled in the cytoplasm, and budding occurs at the outer cell membrane.
- Cleavage of precursor polypeptides into functional structural proteins is mediated by the virus-encoded protease.
DNA Non – enveloped viruses
ADENOVIRUSES
Important Properties –There are 41 known antigenic types; the fiber protein is the main type-specific antigen
- Types 3, 4, 7, and 21 cause respiratory disease
- Types 8 and 19 cause epidemic keratoconjunctivitis
- Types 11 and 21 cause hemorrhagic cystitis
- Types 40 and 41 cause infantile gastroenteritis
Summary of Replicative Cycle
- After attachment to the cell surface via its fiber, the virus penetrates and uncoats, and the viral DNA moves to the nucleus.
- Host cell DNA-dependent RNA polymerase transcribes the early genes, and splicing enzymes remove the RNA representing the introns, resulting in functional mRNA. (Note that introns and exons, which are common in eukaryotic DNA, were first described for adenovirus DNA.)
- Early mRNA is translated into nonstructural proteins in the cytoplasm.
- After viral DNA replication in the nucleus
- Late mRNA is transcribed and then translated into structural virion proteins.
- Viral assembly occurs in the nucleus, and the virus is released by lysis of the cell, not by budding.
Pathogenesis & Immunity- Virus preferentially infects epithelium of respiratory tract and eyes. Adenoviruses infect the mucosal epithelium of several organs (e.g., the respiratory tract [both upper and lower], the gastrointestinal tract, and the conjunctivas). Immunity based on neutralizing antibody is type-specific and lifelong.
In addition to acute infection leading to death of the cells, adenoviruses cause a latent infection, particularly in the adenoidal and tonsillar tissues of the throat.
PAPILLOMAVIRUSES
Important Properties –
Two of the early genes, E6 and E7, are implicated in carcinogenesis. They encode proteins that inactivate proteins encoded by tumor suppressor genes in human cells (e.g., the p53 gene and the retinoblastoma [RB] gene, respectively).
Inactivation of the p53 and RB proteins is an important step in the process by which a normal cell becomes a cancer cell.
Pathogenesis & Immunity
Papillomaviruses infect squamous epithelial cells and induce within those cells a characteristic cytoplasmic vacuole. These vacuolated cells, called koilocytes, are the hallmark of infection by these viruses. Most warts are benign and do not progress to malignancy. However, HPV infection is associated with carcinoma of the uterine cervix and penis
Both cell-mediated immunity and antibody are induced by viral infection and are involved in the spontaneous regression of warts. Immunosuppressed patients (e.g., AIDS patients) have more extensive warts, and women infected with HIV have a very high rate of carcinoma of the cervix.
PARVOVIRUSES
Summary of Replicative Cycle
- After adsorption to host cell receptors, the virion penetrates and moves to the nucleus, where replication occurs.
- The single-stranded genome DNA has “hairpin” loops at both of its ends that provide double-stranded areas for the cellular DNA polymerase to initiate the synthesis of the progeny genomes.
- The viral mRNA is synthesized by cellular RNA polymerase from the double-stranded DNA intermediate.
- The progeny virions are assembled in the nucleus. Released via cell lysis
- B19 virus replicates only when a cell is in S phase, which explains why the virus replicates in red cell precursors but not in mature red cells.
Pathogenesis – B19 virus infects primarily two types of cells: red blood cell precursors (erythroblasts) in the bone marrow, which accounts for the aplastic anemia, and endothelial cells in the blood vessels, which accounts, in part, for the rash associated with erythema infectiosum
Infection provides lifelong immunity against reinfection.
DNA Enveloped Viruses
HERPES VIRUSES
The herpes virus family contains six important human pathogens:
- Herpes simplex virus type 1
- Herpes simplex virus type 2
- Varicella zoster virus
- Cytomegalovirus
- Epstein-barr virus
- Human herpes virus 8 (the cause of kaposi’s sarcoma)
They replicate in the nucleus, form intranuclear inclusions, and are the only viruses that obtain their envelope by budding from the nuclear membrane. The virions of herpesviruses possess a tegument located between the nucleocapsid and the envelope. This structure contains regulatory proteins, such as transcription and translation factors, which play a role in viral replication.
Herpesviruses are noted for their ability to cause latent infections. In these infections, the acute disease is followed by an asymptomatic period during which the virus remains in a quiescent (latent) state. When the patient is exposed to an inciting agent or immunosuppression occurs, reactivation of virus replication and disease can occur
Three of the herpesviruses, herpes simplex virus types 1 and 2 and varicella-zoster virus, cause a vesicular rash, both in primary infections and in reactivations. The other two herpesviruses, cytomegalovirus and Epstein-Barr virus, do not cause a vesicular rash.
Certain herpesviruses are suspected of causing cancer in humans (e.g., Epstein-Barr virus is associated with Burkitt’s lymphoma and nasopharyngeal carcinoma, and human herpesvirus 8 is associated with Kaposi’s sarcoma).

| HSV | VZV | |
| Incubation | 3-6 days | 2 weeks |
| Prodromal symptoms | Burning, itching | Fatigue, hyperesthesia |
| Pain | Moderate | Severe |
| Distribution | Localized | Dermatomes |
| Disease course | Rapid healing | Post herpetic neuralgia in 15% |
| Recurrence | Frequent | Rare |
| Trigger | – Pyrexia – Menstruation – UV/Sunlight – Stress – Malignancy – Old age – Immunosuppression | – Cytotoxics – Dental treatment – Radiation – Alcohol abuse – Malignancy – Old age – Immunosuppression |
| Management | – Acyclovir and analogs – Systemic treatment | – Calamine lotion – Symptomatic |
HERPES SIMPLEX VIRUSES – HSV
Herpes simplex virus type 1 (HSV-1) and type 2 (HSV-2) are distinguished by two main criteria:
– Lesions caused by HSV-1 (primary herpetic gingivostomatitis) are, in general, above the waist, whereas those caused by HSV-2 (genital herpes)are below the waist.
Characteristics—Cross-reaction with HSV-1/2 occurs. No herpes group–specific antigen.
Summary of Replicative Cycle (same for others)
- The HSV has a large double stranded DNA genome, a protein capsid and phospholipid membrane with glycoproteins on the surface
- Viral envelope incorporates into the host cell membrane, the nucleocapsid uncoats and releases the DNA into the nucleus
- In the nucleus, the DNA circularizes and the host enzymes begin to transcribe the first set of viral genes, early genes. The early mRNA molecules enter the cytoplasm, and are translated into early proteins.
- Early proteins enter nucleus where they are required for Viral DNA replication
- Late mRNA transcribed, enters cytoplasm for translation into late proteins aka capsid structural proteins which enter the nucleus
- The late proteins also include envelope proteins which are produced in the ER and become incorporated in the nuclear membrane
- Viral DNA continues to replicate with end to end copies, which are cut into individual genome and packaged into capsids
- Nucleocapsids bud off at nuclear membrane (which has enveloped proteins incorporated in it), through ER and buds off from ER in a vesicle. Migrates to the plasma membrane, fusion of membranes and virus released
http://www.sumanasinc.com/webcontent/animations/content/herpessimplex.html
In latently infected cells, such as HSV-infected neurons, circular HSV DNA resides in the nucleus and is not integrated into cellular DNA. Transcription of HSV DNA is limited to a few latency-associated transcripts (LATS). These noncoding, regulatory RNAs suppress viral replication.
Etiology—Direct contact (contagious)
Pathogenesis & Immunity
Spreads by direct contact, incubation few days to a week, systemic symptoms are fever and malaise. Primary disease course runs for 7-10 days and heals with out scarring.
Secondary disease reactivated by sunlight, stress, immunosuppression. Prodromal symptoms are tingling and burning. Vesicles form superficial ulcer. Heals in 1-2 weeks without scarring.
The virus replicates in the skin or mucous membrane at the initial site of infection
Then migrates up the neuron by retrograde axonal flow and becomes latent in the sensory ganglion cells.
In general, HSV-1 becomes latent in the trigeminal ganglia, whereas HSV-2 becomes latent in the lumbar and sacral ganglia.
During latency, most viral DNA is located in the cytoplasm rather than integrated into nuclear DNA. The virus can be reactivated from the latent state by a variety of inducers (e.g., sunlight, hormonal changes, trauma, stress, and fever), at which time it migrates down the neuron and replicates in the skin, causing lesions.
The typical skin lesion is a vesicle that contains serous fluid filled with virus particles and cell debris. When the vesicle ruptures, virus is liberated and can be transmitted to other individuals. Multinucleated giant cells are typically found at the base of herpesvirus lesions.
Histology—Superficial smear – stain with PAP/Giemsa stain
- Virus infected multinucleated cells
- Intranuclear inclusions (Cowdry type A)
- Intracellular inclusions (Lipschutz bodies)
- Tzanck cells (ballooning degeneration)
Complications:
- Herpetic whitlow – inoculation of virus into finger – pain, redness and vesiculobullous eruptions
- Immunocompromised patient – more chronic and destructive lesions
VARICELLA ZOSTER VIRUS – VZV
Primary disease—Varicella, chicken pox Picture
- Children
- Self limiting
- Clinical:
- Vesicular eruptions of trunk, head and neck – occurs in crops
- Fever, malaise
Secondary disease—Zoster, shingles Picture
- Adults
- Self limiting
- AIDS defining illness
- Clinical:
- Unilateral rash/vesicles along a dermatome
- Post herpetic pain – severe and in 15%
NB: Ramsay Hunt Syndrome – Zoster infection of geniculate ganglion
Pathogenesis—Varicella: Droplet/direct infection. 2 weeks incubation. Initial infection is in the oropharynx. It spreads via the blood to the internal organs such as the liver and then to the skin. Vesicular eruptions on head, neck and trunk – pustular and ulcerate. Fever and malaise. Self limiting with recovery. After the acute episode of varicella, the virus remains latent in the sensory ganglia and VZV DNA is located in the nucleus and is not integrated into cellular DNA. Zoster: Later in life, frequently at times of reduced cell-mediated immunity or local trauma, the virus is reactivated and causes the vesicular skin lesions and nerve pain of zoster. Precipitating factors include leukemia, lymphoma, trauma, radiation. Prodromal symptoms are pain and paresthesia. Vesicular skin eruptions – pustular, ulcerates, dermatomal, unilateral. 15% post herpetic neuralgia
CYTOMEGALOVIRUS – CMV
Pathogenesis—Initial infection usually in the oropharynx. In fetal infections, the virus spreads to many organs (e.g., central nervous system and kidneys). In adults, lymphocytes are frequently involved. A latent state occurs in monocytes. Disseminated infection in immunocompromised patients can result from either a primary infection or reactivation of a latent infection.
Transmitted as:
- IU
- Perinatal
- Breast milk
- Respiratory droplets
- Sexually
- Parentally
EPSTEIN–BARR VIRUS – EBV
Important Properties — The most important antigen is the viral capsid antigen (VCA), because it is used most often in diagnostic tests. EBV enters B lymphocytes at the site of the receptor for the C3 component of complement. Selective binding to CD21 on epithelium and B cells.
Pathogenesis—Infection begins in the pharyngeal epithelium, spreads to the cervical lymph nodes, then travels via the blood to the liver and spleen. EBV establishes latency in B lymphocytes.
Associated with Burkitt’s lymphoma and hairy leukoplakia.
HUMAN HERPESVIRUS 8 – KAPOSI’S SARCOMA–ASSOCIATED HERPESVIRUS
HHV-8, or Kaposi’s sarcoma–associated herpesvirus (KSHV), may be the cause of Kaposi’s sarcoma (KS), the most common cancer in patients with AIDS.
POXVIRUSES
SMALLPOX VIRUS
Important Properties – DNA-dependent RNA polymerase in virion. This enzyme is required because the virus replicates in the cytoplasm and does not have access to the cellular RNA polymerase, which is located in the nucleus.
Summary of Replicative Cycle
- After penetration of the cell and uncoating
- The virion DNA-dependent RNA polymerase synthesizes early mRNA, which is translated into early, nonstructural proteins, mainly enzymes required for subsequent steps in viral replication.
- The viral DNA then is replicated, after which late, structural proteins are synthesized that will form the progeny virions.
- The virions are assembled and acquire their envelopes by budding from the cell membrane as they are released from the cell.
- Note that all steps in replication occur in the cytoplasm, which is unusual for a DNA virus.
Pathogenesis – Smallpox begins when the virus infects the upper respiratory tract and local lymph nodes and then enters the blood (primary viremia). Internal organs are infected; then the virus reenters the blood (secondary viremia) and spreads to the skin. These events occur during the incubation period, when the patient is still well. The rash is the result of virus replication in the skin, followed by damage caused by cytotoxic T cells attacking virus-infected cells.
Immunity following smallpox disease is lifelong; immunity following vaccination lasts about 10 years.
Virology Tables – DNA and RNA Viruses
Below, in table format, is information about viruses classified into their DNA/RNA nuclear structure, nucleocapsid shape, single stranded or double stranded, positive or negative polarity, polymerase, serotype and vaccine available for viruses.
The second table consists the clinical presentation, lab diagnosis and treatment of all types of viruses.





